Clinical particulars
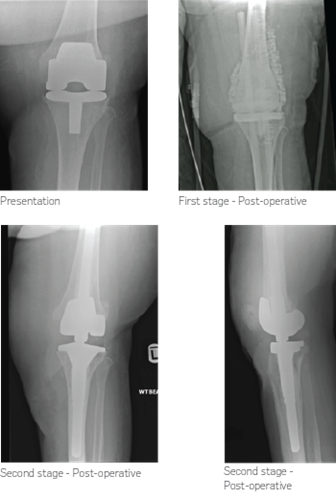
59-year-old female patient presented with infected well fixed total knee replacement two years after primary total knee replacement surgery. This was an acute presentation with all clinical features of infection with samples testing positive for Group B
Streptococcus.
Treatment
First stage – radical debridement, implant removal and insertion of antibiotic-loaded cement spacer and STIMULAN mixed with vancomycin was used to fill the dead space.
Second stage – At 10 weeks, clinically soft tissues were healthy and intra-operative specimens were clear for organisms. Rotating hinge prosthesis was re-implanted.
Outcome
At 1 year follow-up the patient remains infection free and is under regular follow-up.
Note: See STIMULAN page for regulatory statements
.
MA0076R5
