Clinical history
67-year-old male with a history of Type II diabetes, peripheral vascular disease (PVD), hypothyroidism and chronic obstructive pulmonary disease (COPD).
He had a left superficial femoral artery (SFA) angioplasty and stent 2 years previously. He was also scheduled for a left sided downstream SFA/POP (popliteal artery) the following year, however, at the time his foot was stable, and this was cancelled due to the COVID-19 pandemic.
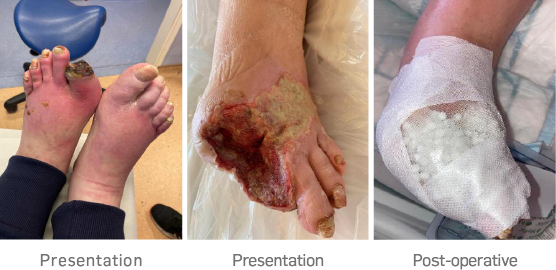
Patient was admitted with chronic limb-threatening Ischemia (CLTI) of the left foot, and necrotic left 1st and 2nd toes, extending to the dorsum of the foot. He also had worsening rest pain.
Treatment
A left leg SFA to posterior tibial artery (PTA) reversed native vein bypass was carried out, followed by 1st to 2nd toes amputation on the left foot with dorsal necrotic tissue debrided. Topical negative pressure dressing was applied post-op, but the patient found this very painful so this was removed.
Deep bone samples sent for microbiology grew Enterobacter cloacae sensitive to co-trimoxazole, gentamicin, amikacin, meropenem and ciprofloxacin.
10cc
STIMULAN beads mixed with antibiotic were packed into the wound cavity and onto the dorsum of the thick biofilm wound layer. The beads were sealed in with Adaptic Touch™, non-adhering silicone contact layer, and mefix tape. A secondary layer of sterile gauze, Soffban
® and crepe bandages applied to manage the exudate levels.
Outcome
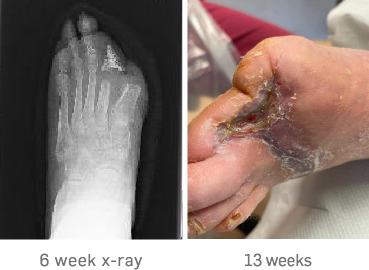
The 6-week x-ray showed healthy bone mineralisation and the bone cortex was intact.
At 13 weeks, the wound had completely healed in a considerably rapid time frame. Post-healing the patient had an uneventful angioplasty for a stenosis to the bypass graft to maintain arterial flow.
This was a very good outcome for a patient with critical limb ischaemia, necrotic tissue loss and a tracking infection.
Had these interventions not happened in a timely manner he would have most likely lost his leg. The patient was very pleased with the outcome and grateful that he did not have to go home with the uncomfortable topical negative pressure dressing.
Note: See STIMULAN page for regulatory statements.
MA0414R1

