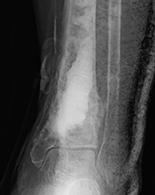
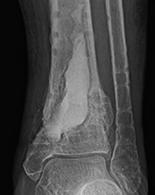
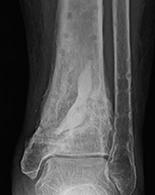
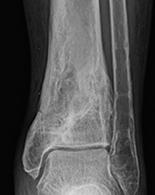
1. Biocomposites Ltd, STIMULAN Instructions for Use. 2. Cooper, J.J., Method of producing surgical grade calcium sulphate; Patent. 1999. 3. Somasundaram, K., et al., Proximal humeral fractures: the role of calcium sulphate augmentation and extended deltoid splitting approach in internal fixation using locking plates. Injury, 2013. 44(4): p. 481-7. 4. Lei, D., et al., Treatment of Distal Radius Bone Defects with Injectable Calcium Sulphate Cement., in Bone Grafting, A. Zorzi, Editor. 2012, InTech. p. 125-134. 5. Lei, D., L. Jing, and S. Yang-yong, Calcium sulfate versus calcium phosphate in treating traumatic fractures. JOURNAL OF CLINICAL REHABILITATIVE TISSUE ENGINEERING RESEARCH., 2008. 12. 6. Lei, D., Z. Ma, and X. Jing, Treatment of bone defect with injectable calcium sulfate powder in distal fractures of radius. Chinese Journal of Bone Tumor and Bone Disease, 2007. 7. Aiken, S.S., J.J. Cooper, and S. Zhou, Osseointegration of a calcium sulphate bone substitute in a large animal model, in The 5th International Congress of Chinese Orthopaedic Association. 2010: Chengdu, China. 8. Lazarou, S.A., G.B. Contodimos, and I.D. Gkegkes, Correction of alveolar cleft with calcium-based bone substitutes. J Craniofac Surg, 2011. 22(3): p. 854-7. 9. Biocomposites, Data on file. 10. Cowie, R.M., et al., Influence of third-body particles originating from bone void fillers on the wear of ultra-high-molecular-weight polyethylene. Proc Inst Mech Eng H, 2016. 230(8): p. 775-83. 11. Cowie, R.M., et al., The Influence of Third Body Damage by a Calcium Sulfate Bone Void Filler on the Wear of Total Knee Replacements., in Orthopaedic Research Society Annual Meeting. 2016: Orlando, FL. p. 103. 12. Lewicki, K., et al., The Effect of Absorbable Calcium Sulfate on Wear Rates in Ultra-high-Molecular-weight Polyethylene: Potential Implications for Its Use in Treating Arthroplasty Infections. J Am Acad Orthop Surg, 2017.
For indications, contraindications, warnings and precautions see Instructions for Use. Concurrent use of locally administered antibiotics may affect setting time.
This brochure may include the use of STIMULAN or techniques that go beyond the current clearance/approval granted by the relevant regulatory authority. Please contact your local representative for further information.
©2024, Biocomposites, STIMULAN, Bringing Calcium to Life, Power to Transform and DRy26 are trademarks/registered trademarks of Biocomposites Ltd. All rights reserved. No unauthorized copying, reproduction, distributing or republication is allowed unless prior written permission is granted by the owner, Biocomposites Ltd.
Patents granted: GB2367552, EP 1204599 B1, US 6780391, EP 2594231 B1, US 8883063, CN ZL201210466117.X, GB2496710, EP 3058899 B1, US 10390954, US 10,588,748, CN ZL201610089710.5
Patents pending: GB1502655.2, GB1704688.9, EP 18275044.8, US 15/933936, CN 108619579A
MA0123R8/MA0246R3/MA0149R3