Clinical particulars
50-year-old female presented with bilateral high energy fractures due to a motor vehicle accident with prolonged extrication. The patient suffered from right open comminuted distal femoral fracture and tibial plateau fracture and left closed comminuted femoral shaft fracture.
Treatment
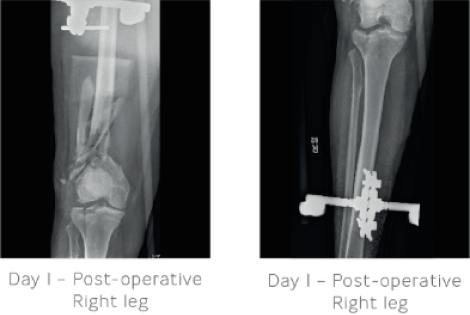
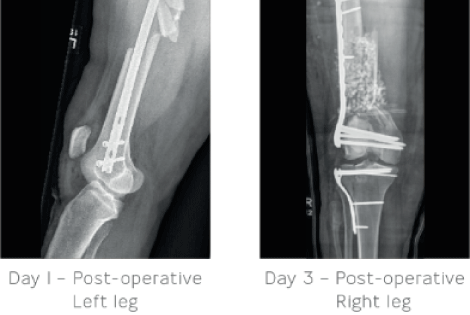
Day 1: Irrigation and debridement of the right femur was carried out along with placement of a large external fixator bridge for temporary stabilisation. The left femur was stabilised using a retrograde IM nail locked statically with screws.
Day 3: The external fixator bridge was removed and open reduction internal fixation was performed on the distal right femur. STIMULAN, cancellous allograft and demineralised bone were used to fill a significant bone deficit just above the femoral condyles. Antibiotic treatment was provided to prevent infection. Open reduction internal fixation was carried out for the tibial plateau fracture supplemented with the use of cancellous allograft.
Outcome
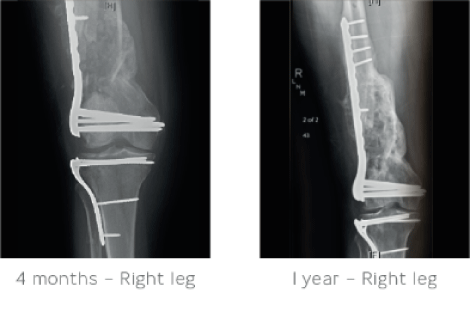
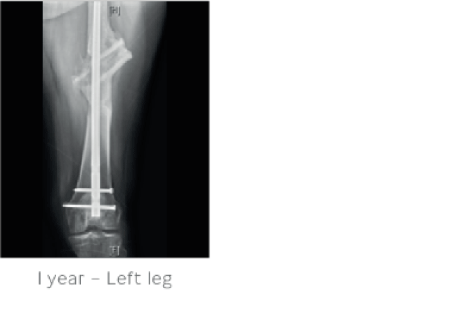
4 months after treatment STIMULAN had absorbed and there was complete healing of the femoral defect. At 1 year follow-up the patient was fully independently ambulatory with a normal gait, normal range of motion and no pain.
Note: See STIMULAN page for regulatory statements
.
MA0181R2



