Clinical particulars
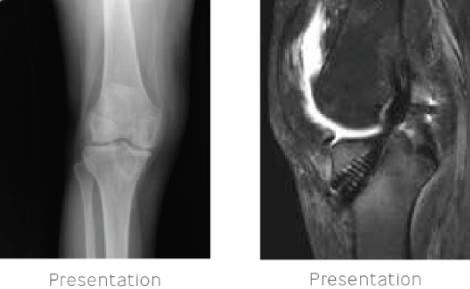
45-year-old female presented with 6 months’ history of knee pain and swelling. This patient had undergone allograft reconstruction of the anterior cruciate ligament 9 months prior. X-ray examination indicated bone tunnel widening and reactive changes in the proximal tibia. MRI examination indicated knee joint effusion and signal changes in the tibia and femur marked the presence of osteomyelitis. Inflammatory markers (ESR/CRP) were elevated and aspiration of the knee joint effusion cultured positive for
Staphylococcus epidermidis.
Treatment
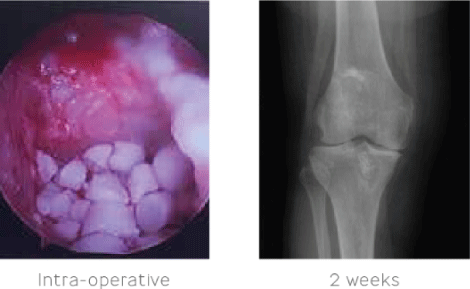
Surgical management was elected with extraction of the allograft. Irrigation and debridement of the tibia and femur were carried out to remove all non-viable tissue with removal of all fixation devices. STIMULAN beads were used to fill the tibia defect and genex® was used to fill the femoral defect. A course of intravenous antibiotics was completed based on the sensitivities of the organism.
Outcome
Pain and swelling resolved post-operatively. Following antibiotic therapy, the infection had cleared clinically 3 months after surgery with normalization of the inflammatory markers and symptomatic improvement.
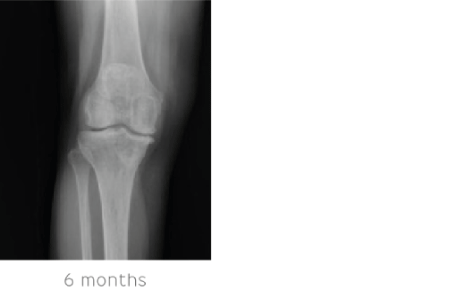
At 6 months’ follow-up both the STIMULAN and genex were fully absorbed and replaced with bone. Inflammatory markers remained normal. The patient had minimal pain secondary to arthritic changes in the knee.
Note: See STIMULAN page for regulatory statements
.
MA0176R2


