Clinical particulars
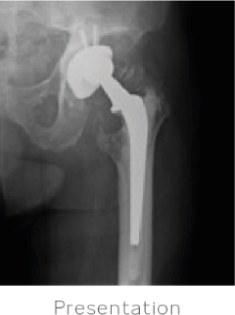
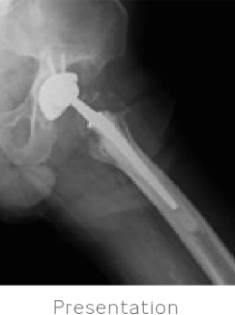
84-year-old male presented with a painful left hip 18 years after a total hip arthroplasty procedure. An extensive examination was conducted to determine the source of the pain. Labs and aspiration were negative for infection. Severe polyethylene wear with extensive resultant osteolysis of the pelvis and proximal femur was identified. A CT scan also demonstrated an unsupportive posterior column with extensive osteolysis of the anterior wall.
Treatment
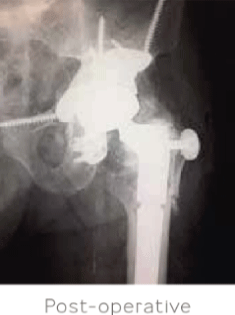
Surgical management was elected with a complex revision of the left THA and pelvic reconstruction using a custom tri-flange cage. STIMULAN beads were used to fill the dead space in the hip and pelvis. Intraoperative cultures were positive for
Enterococcus despite pre-operative examination results. Antibiotic treatment was administered based on the sensitivities of the organism and I.V. antibiotics were continued for 6 weeks following surgery.
Outcome
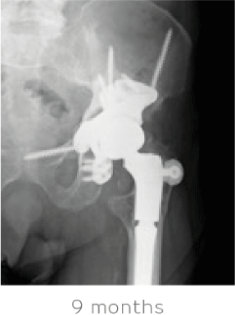
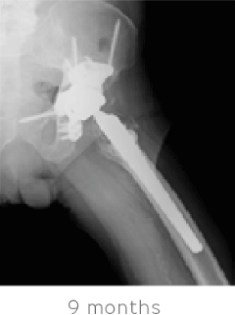
The patient had a successful recovery with well-fixed components, complete absorption of STIMULAN and complete pain relief 9 months after treatment. He remains very active and infection free.
Note: See STIMULAN page for regulatory statements
.
MA0177R2




