Clinical particulars
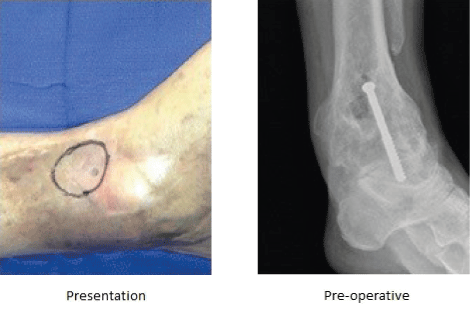
69-year-old male presented with a complex history of his left ankle. The patient was initially treated for a left closed pilon fracture of the distal tibia with open reduction internal fixation. Over the following 13 years, the patient underwent ankle fusion, medial plantar island flap reconstruction due to wound breakdown, and several treatments for wound dehiscence. After an occurrence of wound dehiscence about the medial aspect of the ankle, the patient was hospitalised, an I&D was performed and cultures tested positive for MRSA. The patient underwent weekly wound care visits with daily packing changes at home.
In total, the patient endured 15 surgeries prior to presenting with chronic draining sinus. He was hospitalised and cultures tested positive for
Serratia.
Treatment
Debridement of scarred skin, deep tissue and bone within the chronic osteomyelitis cavity was carried out to remove all non-viable tissue.
STIMULAN mixed with antibiotic was injected as a paste to fill the resulting dead space and treat the bacterial infection. The cavity was covered with a reverse sural flap and I.V. antibiotics were administered.
Outcome
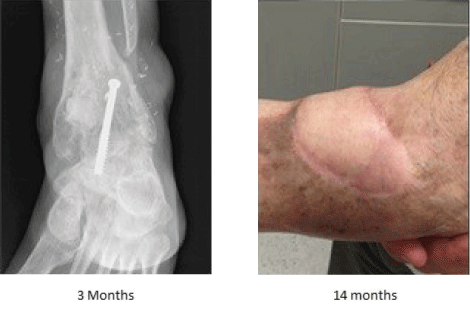
The patient underwent debridement satisfactorily with partial absorption of
STIMULAN paste at 3 months. The patient was fully healed and free from infection 14 months after treatment, and remains infection free 2 years after treatment.
Note: See
STIMULAN page for regulatory statements
.
MA0156R2

